ANNOVERA contraceptive vaginal ring must be inserted before the printed expiration date. Once inserted into the vagina, ANNOVERA will provide contraceptive effectiveness for its full 12-month (13-cycles, so long as it is used according to the directions in the approved prescribing information) even if it expires after insertion. For example, if the expiration date is August 5, 2025, you can insert it on August 4, 2025, and ANNOVERA will remain safe and effective for the entire 13-cycle duration based on clinical studies.
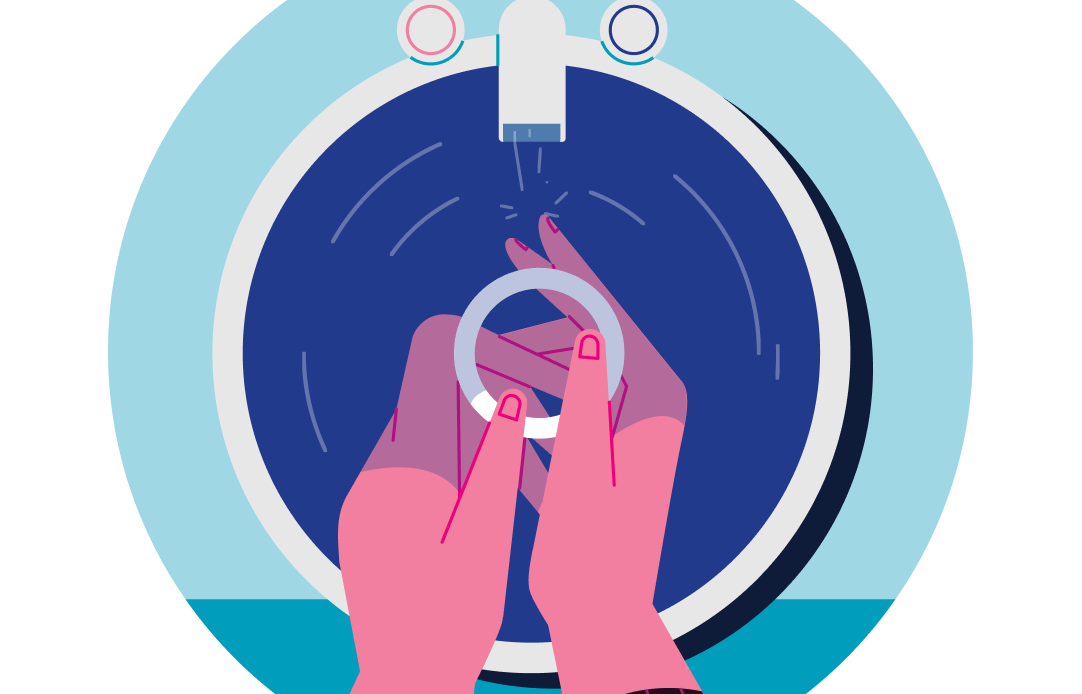
step 1:
clean the ring
First, wash and dry your hands then remove ANNOVERA from the package. Wash the ring with mild soap and lukewarm water. Pat it dry with a clean cloth, towel or paper towel. At the beginning of each cycle, ANNOVERA should be washed and dried this way, before insertion.

step 2:
prepare to insert
Using your thumb and index finger, as shown here, squeeze the ANNOVERA ring into a narrow, oval shape.

step 3:
GET COMFORTABLE
Choose a comfortable position. The ANNOVERA ring can be inserted while lying down, squatting or standing with 1 leg raised on the toilet, bathtub, bed, etc.
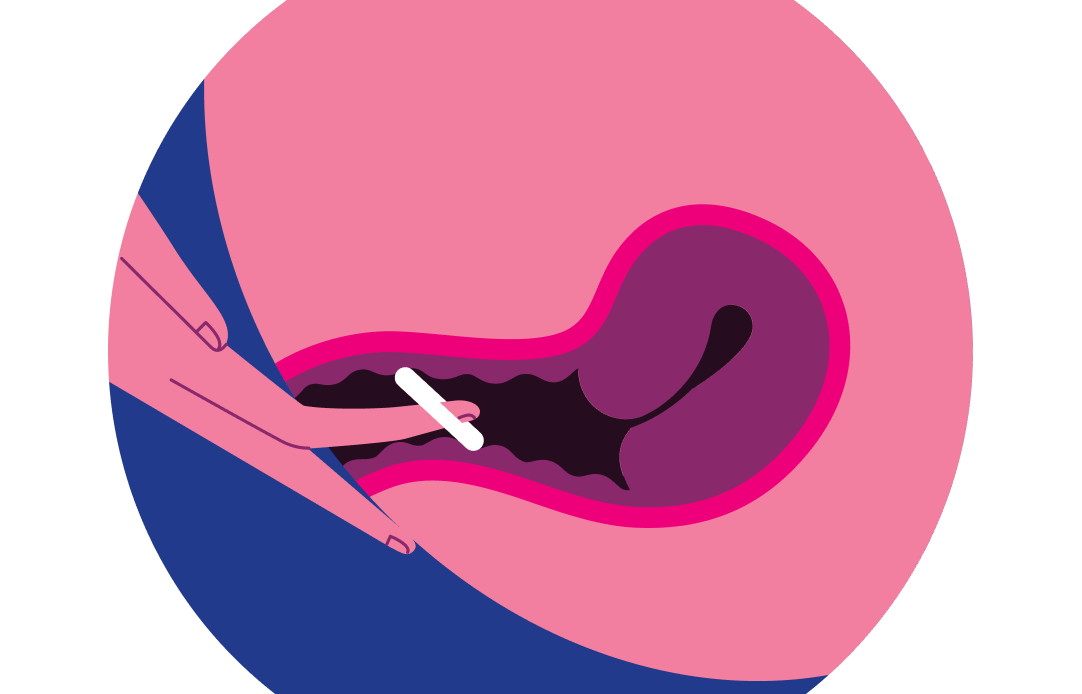
step 4:
inserting annovera
The ANNOVERA ring should be inserted as far as possible into the vagina, and should not be felt. If the ring feels uncomfortable, it may not have been inserted far enough. ANNOVERA does not need to be in an exact position to work. It’s not possible for ANNOVERA to be pushed up too far in the vagina or for it to get lost.
NOTE: Be sure to record the date of insertion. You can also download our ANNOVERA Birth Control Tracker App in the App Store or Google Play.

step 5:
removing annovera
Wash and dry your hands and assume a comfortable position like you did in step 3. Then reach into your vagina, hook the ring with your index finger as shown here, and pull downward and forward to remove it. Wash the ring with mild soap and lukewarm water and pat it dry with a clean paper towel or cloth, and store it in the case provided. Store ANNOVERA away from children, pets and extreme temperatures.
NOTE: Be sure to record the date of removal.
